Safety and Molecular-Toxicological Implications of Cannabidiol-Rich Cannabis Extract and Methylsulfonylmethane Co-Administration.
Study Design
- 研究タイプ
- Other
- 対象集団
- None
- 期間
- 2 weeks
- 介入
- Safety and Molecular-Toxicological Implications of Cannabidiol-Rich Cannabis Extract and Methylsulfonylmethane Co-Administration. 80 mg
- 比較対照
- None
- 主要アウトカム
- None
- 効果の方向
- Neutral
- バイアスリスク
- Unclear
Abstract
Cannabidiol (CBD) is a biologically active, non-psychotropic component of Cannabis sativa whose popularity has grown exponentially in recent years. Besides a wealth of potential health benefits, ingestion of CBD poses risks for a number of side effects, of which hepatotoxicity and CBD/herb-drug interactions are of particular concern. Here, we investigated the interaction potential between the cannabidiol-rich cannabis extract (CRCE) and methylsulfonylmethane (MSM), a popular dietary supplement, in the mouse model. For this purpose, 8-week-old male C57BL6/J mice received MSM-containing water (80 mg/100 mL) ad libitum for 17 days. During the last three days of treatment, mice received three doses of CRCE administered in sesame oil via oral gavage (123 mg/kg/day). Administration of MSM alone did not result in any evidence of liver toxicity and did not induce expression of mouse cytochrome P450 (CYP) enzymes. Administration of CRCE did produce significant (p < 0.05) increases in Cyp1a2, Cyp2b10, Cyp2c29, Cyp3a4, Cyp3a11, Cyp2c65, and Cyp2c66 messenger RNA, however, this effect was not amplified by MSM/CRCE co-treatment. Similarly, no evidence of liver toxicity was observed in MSM/CRCE dosed mice. In conclusion, short-term MSM/CRCE co-administration did not demonstrate any evidence of hepatotoxicity in the mouse model.
要約
Investigation of the interaction potential between the cannabidiol-rich cannabis extract (CRCE) and methylsulfonylmethane (MSM), a popular dietary supplement, in the mouse model found no evidence of hepatotoxicity, and short-term MSM/CRCE co-administration did not demonstrate any evidence of liver toxicity.
Full Text
International Journal of
Molecular Sciences
Article
Safety and Molecular-Toxicological Implications of Cannabidiol-Rich Cannabis Extract and Methylsulfonylmethane Co-Administration
Kristy R. Kutanzi 1, Laura E. Ewing 1,2,3 , Charles M. Skinner 1,3,4, Charles M. Quick 5, Stefanie Kennon-McGill 1,4, Mitchell R. McGill 1,2,4, Larry A. Walker 6,7 , Mahmoud A. ElSohly 6,7,8, Bill J. Gurley 4,6 and Igor Koturbash 1,4,*
- 1 Department of Environmental and Occupational Health, University of Arkansas for Medical Sciences, Little Rock, AR 72205, USA; [email protected] (K.R.K.); [email protected] (L.E.E.); [email protected] (C.M.S.); [email protected] (S.K.-M.); [email protected] (M.R.M.)
- 2 Department of Pharmacology and Toxicology, University of Arkansas for Medical Sciences, Little Rock, AR 72205, USA
- 3 Department of Biochemistry, University of Arkansas for Medical Sciences, Little Rock, AR 72205, USA
- 4 Center for Dietary Supplements Research, University of Arkansas for Medical Sciences, Little Rock, AR 72205, USA; [email protected]
- 5 Department of Pathology, University of Arkansas for Medical Sciences, Little Rock, AR 72205, USA; [email protected]
- 6 National Center for Natural Products Research, University of Mississippi, Oxford, MS 38677, USA; [email protected] (L.A.W.); [email protected] (M.A.E.)
- 7 ElSohly Laboratories, Inc. (ELI), Oxford, MS 38655, USA
- 8 Department of Pharmaceutics and Drug Delivery, School of Pharmacy, University of Mississippi, Oxford, MS 38677, USA
* Correspondence: [email protected]; Tel.: +1-501-526-6638
Received: 28 September 2020; Accepted: 18 October 2020; Published: 21 October 2020
Abstract: Cannabidiol (CBD) is a biologically active, non-psychotropic component of Cannabis sativa whose popularity has grown exponentially in recent years. Besides a wealth of potential health benefits, ingestion of CBD poses risks for a number of side effects, of which hepatotoxicity and CBD/herb-drug interactions are of particular concern. Here, we investigated the interaction potential between the cannabidiol-rich cannabis extract (CRCE) and methylsulfonylmethane (MSM), a popular dietary supplement, in the mouse model. For this purpose, 8-week-old male C57BL6/J mice received MSM-containing water (80 mg/100 mL) ad libitum for 17 days. During the last three days of treatment, mice received three doses of CRCE administered in sesame oil via oral gavage (123 mg/kg/day). Administration of MSM alone did not result in any evidence of liver toxicity and did not induce expression of mouse cytochrome P450 (CYP) enzymes. Administration of CRCE did produce significant (p < 0.05) increases in Cyp1a2, Cyp2b10, Cyp2c29, Cyp3a4, Cyp3a11, Cyp2c65, and Cyp2c66 messenger RNA, however, this effect was not amplified by MSM/CRCE co-treatment. Similarly, no evidence of liver toxicity was observed in MSM/CRCE dosed mice. In conclusion, short-term MSM/CRCE co-administration did not demonstrate any evidence of hepatotoxicity in the mouse model.
Keywords: cannabidiol; cytochrome P450; dietary supplements; hemp extract; hepatotoxicity; herb-drug interaction; methylsulfonylmethane; safety; toxicity
Int. J. Mol. Sci. 2020, 21, 7808; doi:10.3390/ijms21207808 www.mdpi.com/journal/ijms
1. Introduction
Cannabidiol (CBD), a biologically active, non-psychotropic component of Cannabis sativa, has gained significant inroads into the US market over the last year with seemingly endless health claims positioning it as a proverbial panacea for treating stress and pain, boosting energy, enhancing circulation, and even curing arthritis and cancer [1–3]. CBD is a major component of Epidolex®, a drug indicated for the treatment of drug-resistant epileptic seizures associated with some rare pediatric syndromes [4–6]. Furthermore, recent studies indeed indicate significant potential CBD may have on social anxiety disorder and symptomatic improvement in schizophrenia (reviewed in [4–6]) Yet these promises may come at a cost, with a range of adverse side effects noted in vivo and among clinical trials, including diarrhea, vomiting, pneumonia, sedation, and somnolence [4–6]. Furthermore, cardiovascular, neurological, reproductive, embryo-fetal, gastrointestinal, and liver toxicity have been documented subsequent to CBD use at higher doses [5,7–14]. To add to these complications, numerous studies have demonstrated that CBD possesses a significant potential for drug interactions [15–19].
Among side effects associated with higher dose CBD ingestion, the risk for hepatotoxicity is a major concern [20]. For instance, plasma levels of liver aminotransferases were greater than three times the upper limit of normal in patients taking valproate while being treated with Epidiolex®, a purified CBD-containing drug used to treat some rare forms of pediatric refractory epilepsy [16,17,21]. Recent findings have shown that cannabidiol-rich cannabis extract (CRCE) given concomitantly with acetaminophen (APAP), one of the most common over-the-counter medications, exacerbates CBD hepatotoxicity, leading to sinusoidal obstruction syndrome-like liver injury and mortality in the mouse model [15]. These effects were associated with ingestion of high (therapeutic range) doses of CBD, however, the widespread and relatively indiscriminate distribution of CBD-containing products in the supplement market, and the potential inter-individual variability in response to CBD raises concerns as to co-ingestion of CBD and CBD-containing products with prescription and nonprescription drugs.
Dietary supplements (DSs) represent a large group of food items whose regulatory landscape is defined by the Dietary Supplement Health and Education Act (DSHEA 1994). According to DSHEA, pre-market safety assessments for DSs are not required and, while most DSs pose no serious concerns for toxicity, rare cases of herb-induced hepato- and cardiotoxicity as well as clinically relevant herb-drug interactions argue for a more cautious approach to assessing DS safety [22–24].
According to recent polls, up to 70% of U.S. adults took at least one DS within the last 12 months. While it is difficult to realistically assess the current market for CBD and CBD-containing products, their increased popularity suggests that the fraction of U.S. consumers ingesting these products is substantial. Therefore, the probability for concomitant ingestion of CBD with other DSs is very likely, and the potential for CBD–DS interactions necessitates thorough investigation.
In this regard, methylsulfonylmethane (MSM), a natural organosulfur compound, is of particular interest. MSM is found in a wide range of foods, including certain vegetables, fruits, grains, and beverages. Also known as dimethyl sulfone, MSM has become a popular DS, largely for its anti-oxidative and anti-inflammatory properties with some studies indicating that ~10% of supplement users regularly ingest MSM [25]. Numerous patents claiming positive health benefits ranging from stress and pain relief to increased energy and metabolism, enhanced circulation, and improved wound healing [26–29], along with publications showing potential clinical benefit in arthritis and other inflammatory disorders [30,31], have been exploited in marketing of MSM as a popular supplement.
MSM is readily absorbed in the small intestine of both rodents and humans and is characterized by rapid distribution throughout tissues, including the small intestine and liver [32]. Studies indicate that MSM exerts its antioxidant and free-radical scavenging properties by targeting the transcriptional activity of nuclear factor kappa-light-chain enhancer of activated B cells (NF-kB), signal transducers and activators of transcription (STAT), p53, and nuclear factor (erythroid-derived 2)-like 2 (Nrf2) (reviewed in [25]). However, whether or not MSM is capable of affecting cytochrome P450 (CYP)
enzymes in the liver remains unknown. Furthermore, while numerous studies indicate the safety of MSM even at high doses, the potential for MSM-drug/herb interactions remains unexplored.
Therefore, the aim of this study was to investigate whether MSM administration affects the expression of major CYP isoforms and if concurrent use of MSM with CRCE results in MSM–CRCE interactions and/or sensitization to CRCE-induced liver injury.
2. Results
- 2.1. Phytocannabinoid Characterization of CRCE
- 2.2. Anatomical Examination and Physiological Parameters
- 2.3. Histological Findings
Histopathological assessment identified no evidence of necrotic, apoptotic, hemorrhagic, or inflammatory events in experimental mice (Figure 2).
- Figure 1. Physiological parameters in response to methylsulfonylmethane (MSM), CRCE, or MSM/CRCE administration in C57BL6/J mice. (A) Body weight change, (B) liver-to-body weight, (C) kidney-to-body weight, and (D) heart-to-body weight ratios. Data are presented as mean ± SD (n = 6–8). * indicates a significant difference (p < 0.05), and ** indicates a significant difference (p < 0.01), as calculated with a two-way ANOVA and Bonferroni post-hoc test. MSM—methylsulfonylmethane, CRCE—cannabidiol-rich cannabis extract.
- Figure 2. Effects of MSM, CRCE, or MSM/CRCE on liver morphology. (A) vehicle (sesame oil); (B) MSM; (C) CRCE; (D) MSM/CRCE. Magnification: ×40. MSM—methylsulfonylmethane, CRCE—cannabidiol-rich cannabis extract.
- 2.4. Clinical Biochemistry
- 2.5. Cytochrome P450 Expression
Many compounds, regardless of their effect on liver morphology, can have major implications on liver function. To determine if MSM alone or in combination with CRCE could affect the expression of liver drug metabolizing enzymes at administered doses, we evaluated a panel of mouse CYP isoforms (Figure 4).
MSM alone did not affect the hepatic expression of any of the nine investigated CYPs. However, consistent with previous findings, administration of CRCE for three consecutive days increased mRNA levels for seven CYP genes, with Cyp2b10 being most affected (~eight-fold increase) (Figure 4). Interestingly, no induction of Cyp2e1 was observed, an isoform previously reported to be up-regulated shortlyafterCRCEadministration[13]. TheexpressionofCyp2d22, anisoformconsiderednon-inducible, remained unaffected.
Co-administration of MSM with CRCE produced no additional changes in mRNA, as no significant differences were observed between the CRCE and MSM/CRCE experimental groups. These findings suggest that several key Phase I metabolic pathways in the liver may be compromised by CRCE, however, MSM itself does not affect hepatic CYP expression nor does it influence CRCE-associated CYP induction.
- 2.6. Glutathione Measurement
Finally, we evaluated the effect of MSM/CRCE co-administration on the synthesis of hepatic glutathione. The toxicity of xenobiotics is buffered by glutathione, which plays a critical role in protecting cells from both oxidative damage and electrophilic xenobiotic metabolites. Previous studies have indicated that MSM can increase hepatic glutathione synthesis, while administration of high
CRCE doses were associated with pro-oxidative responses [13,34]. Therefore, it was important to evaluate how co-administration of CRCE with MSM might affect glutathione synthesis.
Consistent with previous findings, administration of MSM resulted in modest increases in glutathione, while CRCE alone produced slight reductions in total intrahepatic glutathione (Figure 5A). Furthermore, total glutathione was nearly two-fold higher in MSM/CRCE mice compared to CRCE alone, suggesting the CRCE did not negatively regulate MSM-associated induction of intrahepatic glutathione synthesis. Also, several key enzymes involved in different stages of glutathione synthesis (e.g., Gclm, Gpx1, and Gsr) were unaffected by concomitant ingestion of MSM and CRCE (Figure 5B–D).
3. Discussion
CBD and CBD-containing products are currently on the rise throughout healthcare markets worldwide. Despite its high popularity among consumers, knowledge regarding the negative health effects of CBD-containing products remains limited. Accumulating evidence, however, indicates that CBD, when administered at clinically relevant doses, possesses significant potential for hepatotoxicity as well as for interactions with various conventional medications. In our previous studies, we have investigated the hepatotoxic potential of CRCE, either alone or when co-administered with another potent hepatotoxicant—APAP [13,15]. Therefore, the present study was designed to explore the potential interaction between CRCE and MSM—a popular DS on the U. S. market—that, due to their widespread usage, may overlap in the treatment of various ailments.
Ingestion of MSM itself resulted in no overt toxicological responses, including hepatotoxicity, among experimental mice. This finding is in line with previously conducted studies as well as
its Generally Recognized as Safe (GRAS) status attained in 2007 [35]. Similarly, there was no histopathological evidence of liver injury in mice gavaged with CRCE, nor with the combination of MSM and CRCE as determined by plasma aminotransferases, bilirubin, and miR-122.
At the same time, administration of CRCE or CRCE/MSM caused a significant increase in the liver-to-body weight ratio in mice. This corroborated the findings of other animal studies examining the effects of CBD or CRCE alone [12,13], or in combination with other hepatotoxicants [15]. Xenobiotic-induced increase in liver weight and liver-to-body weight ratio is not a rare finding in rodent models and is not necessarily a prerequisite of liver injury (especially, when other more reliable markers of liver injury are absent) and may be considered an adaptive response. Such adaptive responses may, in turn, be triggered by robust induction of drug metabolizing enzymes, especially Phase I enzymes such as CYPs [36]. Indeed, in this study, CRCE caused a significant increase in the mRNA levels of seven out of nine mouse CYP enzymes. For some like Cyp2b10, which is homologous to human CYP2B6, the increase was substantial, reaching nearly 10-fold. This finding is of particular importance, since tissue harvest occurred 24 h after the last CRCE dose. Taking into account the higher metabolic rates of mice, this suggests CRCE has prolonged effects on hepatic drug metabolizing enzyme activity. Such CYP-associated increases in liver weight are usually adaptive responses, serving a role of metabolism enhancement and detoxification, and often resolve shortly after xenobiotic exposure ceases. The prolonged ingestion of a given xenobiotic may, however, lead to decompensation of adaptive responses and result in hepatotoxicity. Interestingly, most clinical cases of hepatotoxicity associated with the CBD in the form of the drug Epidiolex® were due to its prolonged intake and rarely occurred during the first weeks of treatment [21].
Out of seven CYP enzymes whose expression was induced by CRCE, of particular interest is Cyp2b10, a mouse enzyme homologous to human CYP2B6. In CRCE-treated groups, its expression reached nearly 8–10-fold compared to vehicle-treated or MSM-only animals. This data is in agreement with findings within the B6C3F1 mouse strain, in which Cyp2b10 was the most up-regulated CYP gene after both single and repeated dosing with CRCE [13]. This CYP isoform is known to play a key role in the metabolism of prescription drugs, such as cyclophosphamide, ketamine, and bupropion. Furthermore, CYP2B6 together with CYP3A4 are central to the metabolism of clobazam, a common anti-seizure medication used in the treatment of many forms of epilepsy. Here, we demonstrate that CRCE, besides eliciting a robust upregulation of Cyp2b10 mRNA, increased expression of two CYP3A4 mouse homologs—Cyp3a4 and Cyp3a11. Clinical trials confirm this finding, demonstrating higher serum concentrations of N-desmethylclobazam, the active metabolite of clobazam, when the latter is co-administered with CBD (Epidiolex®) [24]. Though this elevation of N-desmethylclobazam may also be attributable to inhibition of CYP2C19, contributions of CYP3A and CYP2B6 induction have also been recognized [37,38]. This ability to serve as a substrate for multiple CYPs suggests that CBD-containing products may have a significant interaction potential with other drugs besides clobazam.
MSM is rapidly absorbed from the gastrointestinal tracts of both humans and rodents [39]. The exact metabolic fate of MSM is not fully understood, however, its tissue distribution and liver accumulation as well as formation by hepatic microsomes have been reported [40]. Studies in rats further demonstrate that up to 80% of MSM is excreted unchanged [41]. Here, we demonstrated that MSM caused no measurable changes in the expression of a panel of mouse hepatic Cyps. Furthermore, pre-treatment with MSM for two weeks did not affect CRCE-associated CYP induction, suggesting a rather inert nature of MSM in the mouse liver.
Despite the liver’s seemingly minor role in MSM clearance, an indirect potential for an MSM/CRCE interaction cannot be dismissed. For instance, a recent report indicated an important role Cyp2c29, another CYP affected by CRCE in this study, plays in liver injury [42]. This homolog of human CYP2C8 and CYP2C9 was found to be continuously down-regulated during liver injury. In contrast, reversal of Cyp2c29 expression in APAP- and CCl4-induced hepatotoxicity (two common models of liver injury), resulted in suppression of pro-inflammatory signaling and cytokine production by targeting NF-kB pathway [42]. Interestingly, similar anti-inflammatory effects associated with NF-kB
inhibition were reported for MSM [43,44]. These findings may have a significant therapeutic value and further investigations of the MSM/CRCE interaction potential and its effects on NF-kB signaling pathway are clearly warranted.
Previous studies have demonstrated that high doses of CBD/CRCE can deplete intrahepatic glutathione and its synthesis [13,15]. The latter plays a critical role in protecting cells from oxidative damage and the toxicity of xenobiotic electrophiles by maintaining redox homeostasis. MSM has been previously reported to effectively modulate synthesis of glutathione in experimental models [34]. The exact mechanism(s) underlying this modulatory effect remain unknown, but by serving as an alternate source of sulfate for the sulfation and detoxification of various compounds, MSM may spare cysteine for the synthesis of glutathione [45–47]. Therefore, it is critical to understand whether or not the ability of MSM to regulate glutathione will be hampered by concurrent ingestion of other DSs, particularly the popular glutathione-depleting CBD-containing products. Importantly, we demonstrate that mice treated with MSM and subsequently challenged with CRCE exhibited a nearly two-fold increase in hepatic glutathione levels, compared to CRCE alone. Furthermore, the potential of MSM to mitigate toxin-induced liver damage, such as with carbon tetrachloride [48], paraquat [45], or APAP [46], suggests that MSM can serve as a valuable hepatoprotective agent, including in cases of CBD/CRCE-induced liver injury.
In conclusion, we report that concurrent short-term administration of MSM with CRCE is not hepatotoxic in an experimental mouse model. Furthermore, the potential for an MSM/CRCE interaction appears low based upon the lack of CYP modulation by MSM. Lastly, MSM’s ability to enhance hepatic glutathione synthesis was not affected by CRCE, a finding suggestive of its capability for mitigating CBD/CRCE-induced hepatotoxicity. Future studies are warranted to explore this potential, as well as the nature of long-term co-administration of CRCE with other entities.
4. Materials and Methods
- 4.1. CRCE Extract Characterization, Dosing Solution, and Dose Calculations
- 4.2. Animals
Male C57BL6/J (7-weeks of age) were purchased from Jackson Labs (Bar Harbor, MA, USA). Animals were acclimated for 1 week prior to study initiation and randomized into cohorts of vehicle (sesame seed oil) and experimental groups: MSM, CRCE, and MSM/CRCE (n = 6 per group, except for the MSM/CRCE group which contained 8 animals). Each animal was individually identified with an ear tag. Mice were housed 3 per cage (4 animals per cage for the MSM/CRCE group) in polycarbonate cages in an appropriate animal room at the University of Arkansas for Medical Sciences (UAMS) Division of Laboratory Medicine from arrival until euthanasia. Room temperature was maintained at
19–22 ◦C with a relative humidity of 55–70%. Automatic light controls were set to provide fluorescent lighting for a 12 h photoperiod (07:00–19:00 for light phase). All procedures were approved by the UAMS Institutional Animal Care and Use Committee (protocol number: AUP #3902), and all personnel followed appropriate safety precautions.
Mice received MSM (Bergstrom Nutrition, Vancouver, WA, USA) in drinking water (80 mg/100 mL) ad libitum for 17 days. The dose of MSM chosen for this study was based on previous reports [51]. Water intake was measured frequently to ensure dosage of MSM did not differ significantly between animals. Animals in the CRCE groups were gavaged with 123 mg/kg/day CBD (MED of 10/mg/kg/day, a commonly used dose of CBD in clinical settings) on Monday, Tuesday, and Wednesday of Week 3 (days 15 through 17) at 08:00. All animals were scheduled to be terminated 24 h after the last gavage treatment of the CRCE groups. Pelleted rodent diet was available ad libitum except for an overnight fasting period prior to necropsy.
Mice were anesthetized with isoflurane and terminal bleeding was performed by a trained animal technician. Anesthetized animals then underwent cervical dislocation to ensure death and tissues were collected immediately. Animal body, liver, heart and kidney weights were recorded at the time of necropsy. Livers were excised, 1-mm sections were obtained from the left and right lateral lobes, and placed in a tissue cassette for further histological evaluation. The remaining liver tissue was snap-frozen in liquid nitrogen and stored at −80 ◦C for further molecular and biochemical analysis.
- 4.3. Histopathological Assessment
- 4.4. Blood Sampling and Clinical Biochemistry
- 4.5. miR-122 Analysis
- 4.6. Gene Expression
Total RNA was extracted from liver tissue using the RNeasy Mini Kit (QIAGEN, Germantown, MD, USA) according to the manufacturer’s protocol. Following purification, 1000 ng were reverse
transcribed with the High Capacity cDNA Reverse Transcription Kit following the manufacturer’s instructions (Thermo Fisher, Waltham, MA, USA). Primers were added at a final concentration of 5 µM. The list of assays used in this study can be found in Table S1. Gene expression values were normalized to the internal control gene 18S and expressed as fold change according to the ∆∆Ct method.
- 4.7. Glutathione Measurement
- 4.8. Statistical Analysis
All statistical analyses were performed with the Graphpad Prism 6 software (Graphpad Software. San Diego, CA, USA). Groups were compared with a two-way ANOVA followed by a Bonferroni multiple comparison test when appropriate. An adjusted p-value of <0.05 was considered significant.
Supplementary Materials: Supplementary materials can be found at http://www.mdpi.com/1422-0067/21/20/ 7808/s1.
Author Contributions: Conceptualization, B.J.G. and I.K.; methodology, L.E.E., C.M.S., S.K.-M., M.R.M., and M.A.E.; formal analysis, K.R.K., L.E.E., C.M.S., C.M.Q., S.K.-M., M.R.M., L.A.W., M.A.E., B.J.G., and I.K.; resources, M.R.M., M.A.E., L.A.W., B.J.G., and I.K.; writing—original draft preparation, K.R.K., B.J.G., and I.K.; project administration, B.J.G. and I.K.; funding acquisition, I.K. All authors have read and agreed to the published version of the manuscript.
Funding: This research was funded by Bergstrom Nutrition (Vancouver, WA, USA), an Institutional Development Award (IDeA) from the National Institute of General Medical Sciences of the National Institutes of Health under grant number 1P20GM109005; the National Institute of General Medical Sciences, grant number T32GM106999; Clinical and Translational Science Awards, UL1TR000039 and KL2TR000063; and the Arkansas Biosciences Institute.
Acknowledgments: The authors would like to thank Robin Mulkey and Bianca Schutte for providing excellent animal care at the UAMS Animal Facility.
Conflicts of Interest: This study, in part, was funded by Bergstrom Nutrition (Vancouver, WA, USA). The funder had no role in the design of the study, in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results. The authors declare no other potential conflict of interests.
Figures
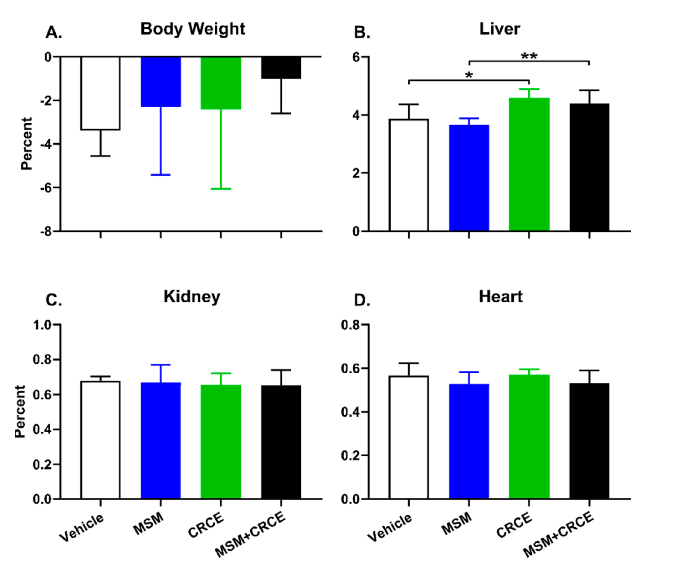
Figure 5
Extended molecular analysis data examining the effects of CBD-rich cannabis extract and MSM on cellular detoxification pathways. The results contribute to understanding the safety implications of co-supplementation.
chart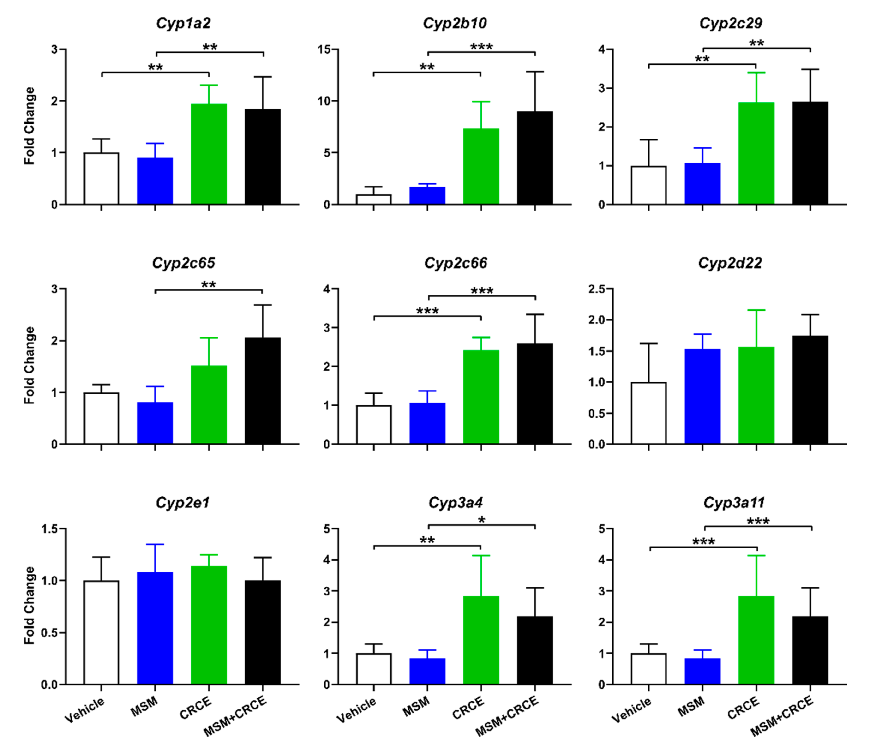
Figure 6
Intrahepatic expression of cytochrome P450 enzymes in response to MSM, CBD-rich cannabis extract (CRCE), or their combination. Alterations in CYP450 expression are critical for assessing drug-supplement interaction potential and metabolic safety.
chart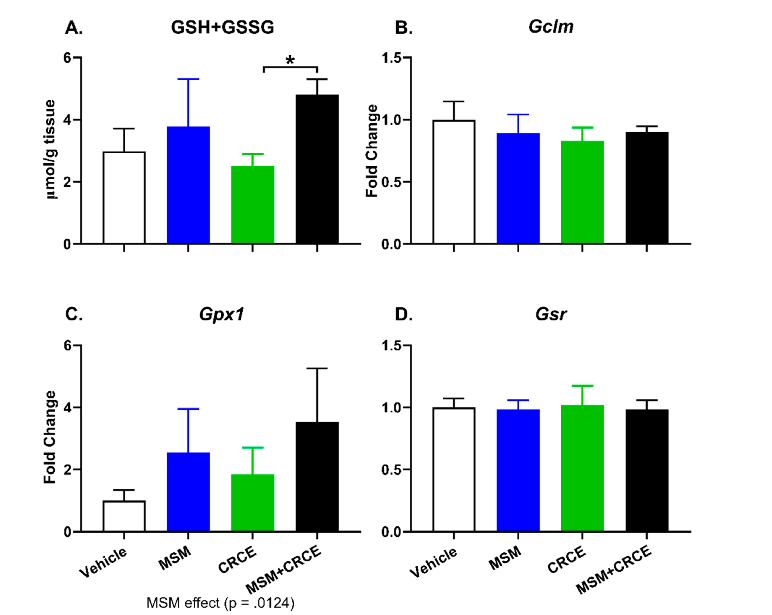
Figure 7
Effects of MSM, CBD-rich cannabis extract, and their combination on intrahepatic glutathione synthesis and related gene expression. Glutathione is a key antioxidant, and changes in its production may indicate shifts in the liver's oxidative stress response.
chartUsed In Evidence Reviews
Similar Papers
Clinical infectious diseases : an official publication of the Infectious Diseases Society of America · 2005
Prevalence of and associated risk factors for fluoroquinolone-resistant Neisseria gonorrhoeae in California, 2000-2003.
Journal of bone and mineral metabolism · 2013
Assessment of safety and efficacy of methylsulfonylmethane on bone and knee joints in osteoarthritis animal model.
Animal bioscience · 2022
Effects of dietary methyl sulfonyl methane and selenium on laying performance, egg quality, gut health indicators, and antioxidant capacity of laying hens.
Microorganisms · 2022
Bioprospecting and Molecular Identification of Used Transformer Oil-Degrading Bacteria for Bioplastics Production.
Alcoholism, clinical and experimental research · 2022
Correlates of high phosphatidylethanol (PEth) levels and their concordance with self-reported heavy alcohol consumption among men who have sex with men who binge drink alcohol.
Journal of behavioral medicine · 2022